If you make a methylester out of the top OH you get codeine with antianalgesic, antitussive and antidiarrheal properties. Adding acetyl to both OH's gives the lipophilic form heroin, a highly addictive drug.
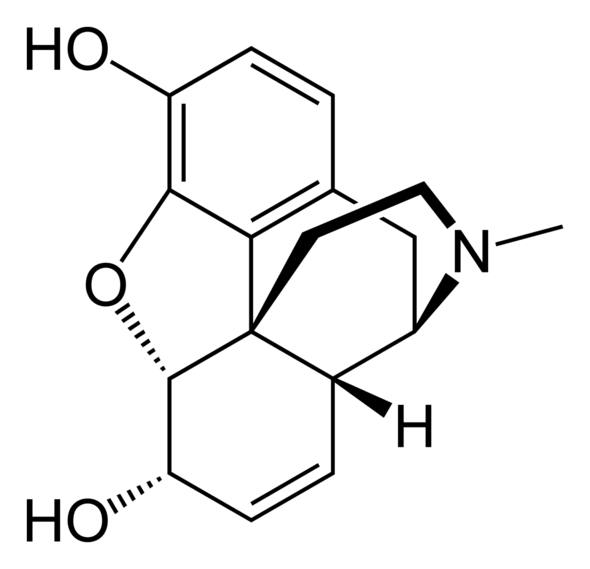
Morphine's structure is fairly complex. You have three condensed rings, an oxygen bridge and a nitrogen bridge. Before you scroll down you can try to guess its structure. Which rings are in the same plane? The key is to consider that the c-hexene ring is annexed in a cis configuration.

The benzene and c-hexane rings and the oxygen bridge are in the same plane. The c-hexene ring and nitrogen bridge are in a plane perpendicular to it.

The ring seen on top might resemble the terminal tyrosine of β-endorphin. Besides that the structure does not really look like the 31 amino acid peptide.


[1] You save o, m, and e. But it is arguable which o is taken out.
I finally found out how I can use javascript in blogspot. This is an interactive model of morphine produced with the help of jmol. Change the view with left and middle mouse buttons. Use right click for options.
Well, jmol does not work anymore on my blog. But since so many people seem to be looking at this post, I'll try TwirlyMol. And if even that does not load, click here.
5 comments:
The Jmol applet is awesome.
Any way I can embed a jmol applet into a powerpoint presentation?? morphine's just not as cool 2D :)
i thought about it but i haven't figured out how to do it and I would not know how to link to the library files.
if you want it rotating you could just create the movie in pymol and show it on the slide. that also seems to be more stable. even though the applet would be more fancy
Any idea where can I find images of morphine enantiomers? I should include them in a presentation and it drives me crazy...
By the way, great ob with the website!
if you just want the enantiomer, you just have to take the mirror image (for example in paint) of any picture of morphine.
for general diastereomers it is not so easy and you would need some molecular modeling basics, I think. you could draw it in avogradro and then optimize it maybe
Post a Comment