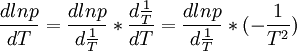It's been quite a while since I've written anything here. I don't know how exciting the topic is that I picked for coming back. But it's something to think about once. In physical chemistry (which is the common favorite of everyone doing chemistry) you often run across terms of the following form:

Now the question is how do we estimate that if we don't have a calculator at hand? And more important how do we do it with all the different units that are around?
We start out with a handy table that gives us the values of kT (at room temperature) in the different units that we are interested in. (Non-energy units are converted using Planck's constant and the speed of light in the usual way.)
| 298 | K |
| 2.478 | kJ/mol |
| 0.592 | kcal/mol |
| 0.026 | eV |
| 0.00094 | Hartree |
| 4.11E-21 | J |
| 207.1 | cm-1 |
| 6.21E12 | Hz |
| 48281 | nm |
Now we can estimate things because we know that:

and

For example we can say that a hydrogen stretch at 4000 cm
-1 will not be active at room temperature because it's energy is much higher than 207 cm
-1 which corresponds to kT. Or we can say that a reaction with a Gibbs free reaction energy below -25 kJ/mol will be almost quantitative.
But to be certain we need the following two formulae which will help to estimate the order of magnitude (ln is the natural logarithm, lg the decadic logarithm)[1]:

and

Now we can do pretty much everything we ever wanted to do.
In the first two examples we see that we are in the order of 10
-4 and the guess was correct.
If we stick to normal modes, we can for example be wondering to what extent a 600 cm
-1 normal mode is in its first excited state. Using the first formula we get:

That's below10% but well above 1% (the exact value is 5.5%). And we know that a mode at 600 cm
-1 is still excited a little bit.
But of course you can also do it for more complex things. For example coalescence in NMR. For an isomerising molecule with activation barrier ΔG coalescence is reached if the following condition is fulfilled:

Δν is the frequency difference between the peaks, e.g. 0.5 ppm of 600MHz which is 300 Hz. kT/h is the frequency term in the Eyring equation, it actually corresponds to my value from above, 6.21E12 Hz, but I think for a different reason [2]. If you divide by it you get:

Apply the logarithm formula and put in the value of kT (or actually RT) and you get:

And that's about the actual value.
[1] The advantage is that you have to remember only to digits 2.3 and you almost get four digit accuracy compared to the exact value 2.302585...
[2] I would say that the value is chosen because it is a typical frequency of a skeletal mode but it is coincidence that this is close to kT.